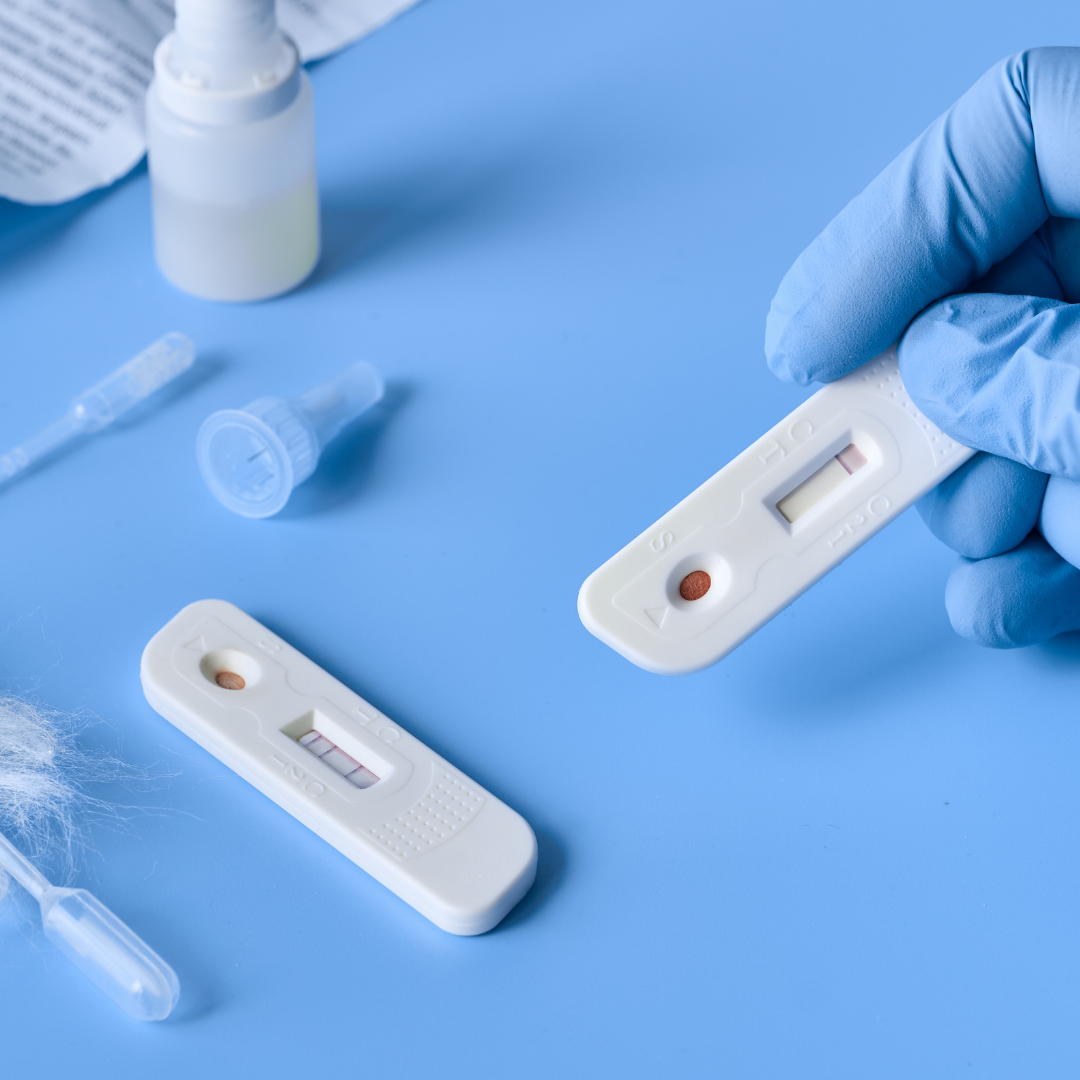
With so much reporting on rapid antigen testing, it is timely to sit back and look at some of the published data on the performance of rapid antigen tests, and conclusions being made by experts about the use and applicability of rapid antigen tests.
In March 2021, Jacqueline Dinnes from the Institute of Applied Health Research, the University of Birmingham along with a number of other authors from that and other institutions completed a review of 22 studies of rapid antigen and molecular-based tests for COVID-19. Their findings were contained in their article “Rapid, point-of-care antigen and molecular-based tests for diagnosis of SARAs-CoV-2 infection”.
In that review, the authors observed that predicted values based on a hypothetical cohort of 1000 people with suspected COVID-19 infection (with a prevalence of 10%) resulted in 105 positive test results including 10 false positives (positive predictive value 90%), and 895 negative results including 5 false negatives (negative predictive value 99%).
Her and her co-authors concluded that in people with signs and symptoms of COVID-19, sensitivities are highest in the first week of symptoms onset when viral loads are higher.
The authors considered that antigen tests which met the World Health Organisation’s ‘target profiles’ for COVID-19 diagnostic products (‘acceptable’ sensitivity ≥ 80% and specificity ≥ 97%), can be considered as a replacement for laboratory-based PCR when immediate decisions about patient care must be made, or where PCR cannot be delivered in a timely manner.
The authors acknowledged some of the limitations of their review of the literature. The early-stage evaluations of point-of-care tests for detecting SARS-CoV-2 infection they had studied were largely based on remnant laboratory samples. They noted that the findings may have limited application to testing performed as part of clinical practice or to asymptomatic cases. Other variables such as the nature of the symptoms of COVID-19 or their duration in infected people may also limit the efficacy of their analysis.
They observed that further evaluations of rapid tests for COVID-19 infection in clinical settings were urgently needed.
Jacqueline Dinnes followed up this article with a piece in The Lancet (17 July 2021) in which she concluded that:
“The ability of RDTs to quickly pick up the majority of individuals with high levels of virus is undeniable but these tests do still miss people who are likely to be infectious and also risk falsely classifying uninfected people as positive.”
She remarked that future studies of rapid antigen test performance may use cohorts that included those “sufficiently motivated to attend and a greater proportion of people with [symptoms] for testing”, and that this may influence the results of those studies.
In June 2021, experts from the Institute of Population Health at the University of Liverpool analysed almost 750,000 tests taken from 283,338 residents in Liverpool, as part of its Covid-SMART community testing initiative between December 2020 and April 2021. This meant over 57% of Liverpool residents took part in the testing.
The testing was conducted on people that did not have symptoms onset – thereby helping fill in one of the blanks from the University of Birmingham review.
Testing was performed in key workplaces, including emergency services and in schools, based on three types of testing: 1. ‘test-to-protect’ vulnerable people and settings (for example, people living in care homes); 2. ‘test-to-release’ contacts of confirmed infected people sooner from quarantine than the stipulated period (for example, key workers in quarantine); and 3. ‘test-to-enable’ careful return to restricted activities to improve public health, social fabric, and the economy (for example, visits to care homes or sports events).
The Innova Medical Group SARS-CoV-2 Antigen Rapid Qualitative Test, using a nasal swab, was used. The test was conducted in the midst of rising cases involving the Alpha variant of COVID-19.
The test was found to detect most COVID-19 cases without classical symptoms but with high viral load.
More specifically, it found that the rapid antigen test returned the same negative result as the PCR test in tested residents in 99.2% of cases (i.e. the negative predictive value), and the same positive test result as the PCR test in 90.3% of cases with a high viral load (i.e. the positive predictive value).
Consequently, the number of positive COVID-19 cases detected rose by approximately 18%, and the number of new positive COVID-19 cases was reduced by approximately 21%.
By way of interest, the Innova Test was subsequently the subject of a warning from the US Food and Drug Administration (FDA) warning to the public to stop using the test on 10 June 2021, due to concerns about the performance of the test, and labeling which included performance claims that did not accurately reflect the performance estimates observed during clinical studies of the tests. Innova recalled the test in the US on 23 April 2021. It remains on sale in the UK and Europe.
The final study covered in this blog is that performed on seven rapid antigen tests available in the European Market by Victor M Corman from the Berlin Institute of Health, Berlin, Germany and his colleagues, the results of which were published in their article in The Lancet in July 2021.
The authors conducted a laboratory test of various rapid antigen tests using a variety of samples including: recombinant SARS-CoV-2 nucleoprotein, cultured endemic and emerging coronaviruses, stored respiratory samples with known SARS-CoV-2 viral loads, stored samples from patients with respiratory pathogens other than SARS-CoV-2, and self-sampled swabs from healthy volunteers.
The authors noted that the tests studied (except for one) did contain “sensitivity range[s]” that “overlap[ped] with viral load figures from the infectious period in most patients”, meaning they were generally sufficiently sensitive to pick up the virus during this phase.
However, they noted that “a steep increase in virus concentration occurs around or before the onset of symptoms”.
Consequently, “Due to the rapid change of viral load in the acute phase of COVID-19, [rapid antigen tests] have a narrow timeframe for their useful application that predominantly comprises the first week of symptoms . . . Screening of asymptomatic patients with the expectation of being able to discern between virus absence and asymptomatic or presymptomatic infection is more difficult.”
Stay tuned for more updates and analysis.